
Your experience matters, make it matter for others
Make a difference by giving feedback on a video game treatment for kids with ADHD targeting inattention.
What’s required of you:
Over the course of six months, complete study activities approximately once a month and get compensated for your involvement. You can stop participating at any time!
If you already signed up within the past 6 months, then there is no need to sign up again.
Important note:
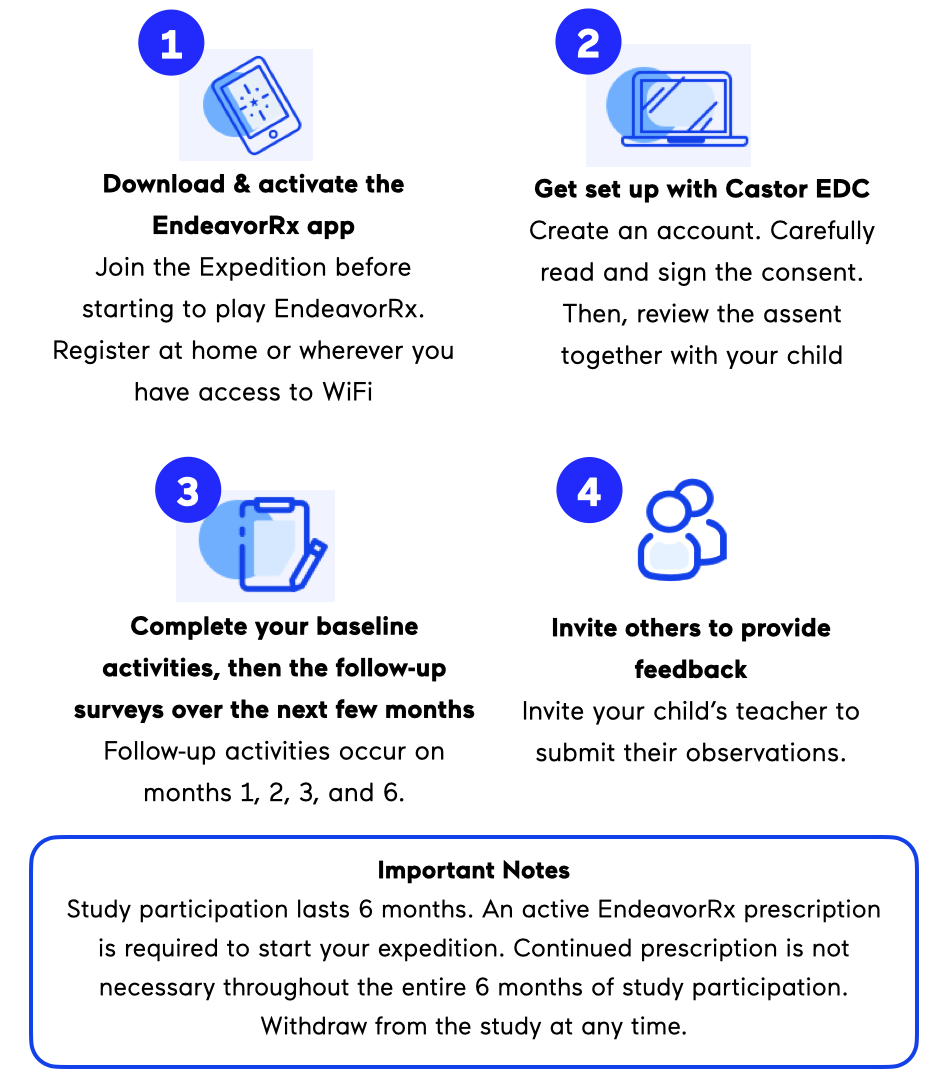
A valid and active EndeavorRx prescription is required to enroll. However, you do not need to keep an active prescription during the entire 6 months of participation. If you enroll without an active EndeavorRx prescription, your submitted data will be deleted upon verification of non-prescription. See our privacy notice at my.akili.care/privacy.
STUDY DETAILS
The purpose of this study is to better understand what happens during and after your child’s EndeavorRx® treatment period, and to understand how EndeavorRx is being used by patients and healthcare providers.
As a participant in this study, you (the caregiver) will be asked to complete study surveys activities approximately once a month for 6 months. As part of the study, you can also invite your child’s teacher to submit surveys of their observations of your child’s ADHD symptoms. All data are captured electronically and study activities can be completed from your home or anywhere you have access to WiFi.
For your participation, you will receive $25 for each month’s study activities you complete. You do not have to complete all six months to receive study compensation, and you can choose to stop participating at any time.
Your decision to participate or not does not affect you and your child’s access to or quality of the EndeavorRx treatment.
HOW TO PARTICIPATE
If you are interested in signing up, enter your email into the field below. You can then expect to receive an email inviting you to create an account with Castor EDC. Castor EDC is an electronic data capture tool used to document your consent and survey responses. You will receive a consent form, through Castor EDC, that provides further details to help you decide whether you and your child would like to participate. Once you have signed the consent form, you will start receiving monthly study surveys.
The information and data collected from you and your child via the study will be used in accordance with the provisions consent form you signed as well as the provisions of Akili’s Privacy Notice.
-
Contribute to the study of a new type of medicine for ADHD
-
Participate fully remotely through online study activities - no doctor’s visits required
-
Complete study activities once a month
-
Receive $25 for every set of monthly study activities you complete
-
Withdraw at any time

